Drug Supply Chain Security Act (DSCSA) Unpacked
Challenges in Pharmaceutical Traceability
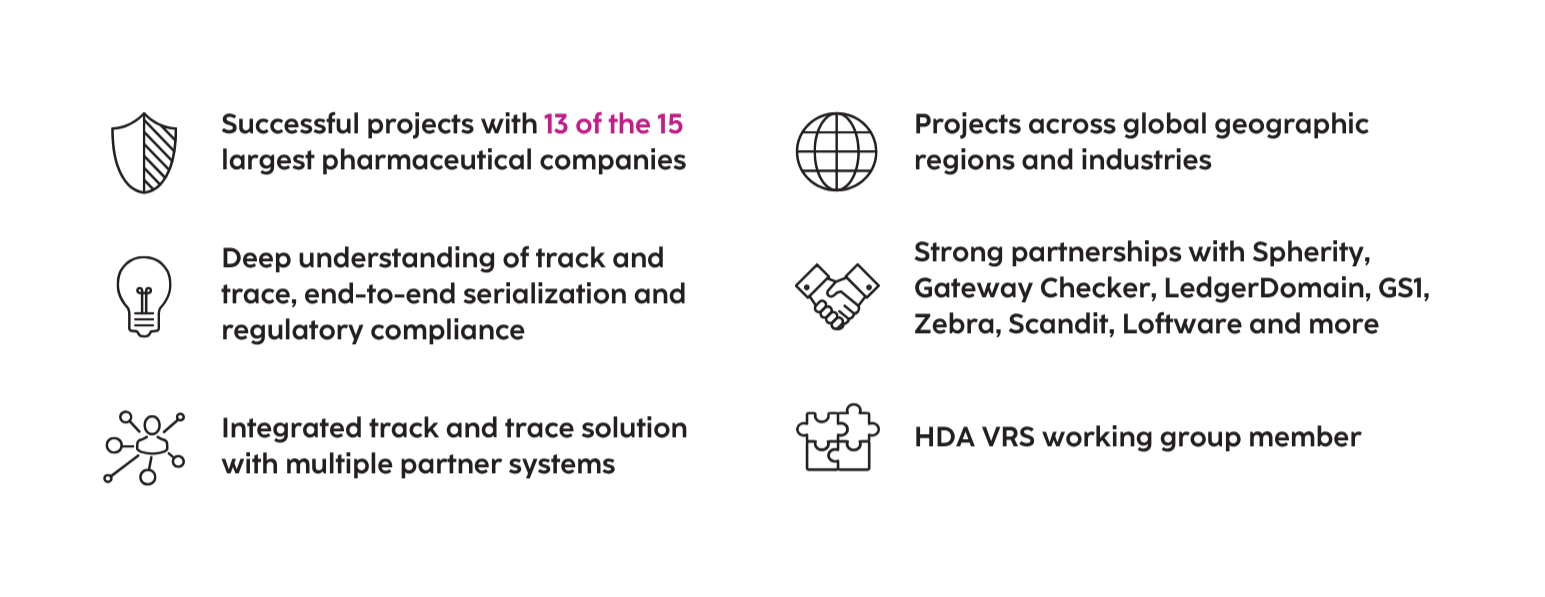
The U.S. pharmaceutical market accounts for over 40% of global pharmaceutical sales and is expected to grow consistently in upcoming years, reaching over $634 billion in 2024.ٟ This rise is driven by a growing, aging population with higher disposable income. Although the vast majority of pharmaceuticals sold in the U.S. are safe, this large and growing market is an attractive target for theft and counterfeiting. A long-standing estimate by the World Health Organization (WHO) suggests that between five and eight percent of pharmaceuticals sold worldwide is counterfeit; many experts consider this estimate low. Counterfeit or falsified medical products can contain the wrong active ingredients, no active ingredients or toxic ingredients, and are often produced in unsanitary conditions by untrained personnel. Furthermore, they are carefully produced to look identical to genuine products, making them difficult to detect. These fraudulent products can either fail to treat a patient's condition or cause a dangerous and possibly fatal reaction. There have been numerous high-profile cases where illegitimate pharmaceuticals have caused illness and death. To protect patients from these dangers, the safety of the life sciences supply chain is essential. Many countries worldwide have already enacted and enforced serialization and Track & Trace requirements. The Drug Supply Chain Security Act (DSCSA) is designed as a tool to help combat illegitimate pharmaceutical products in the U.S.
About the Drug Supply Chain Security Act (DSCSA)
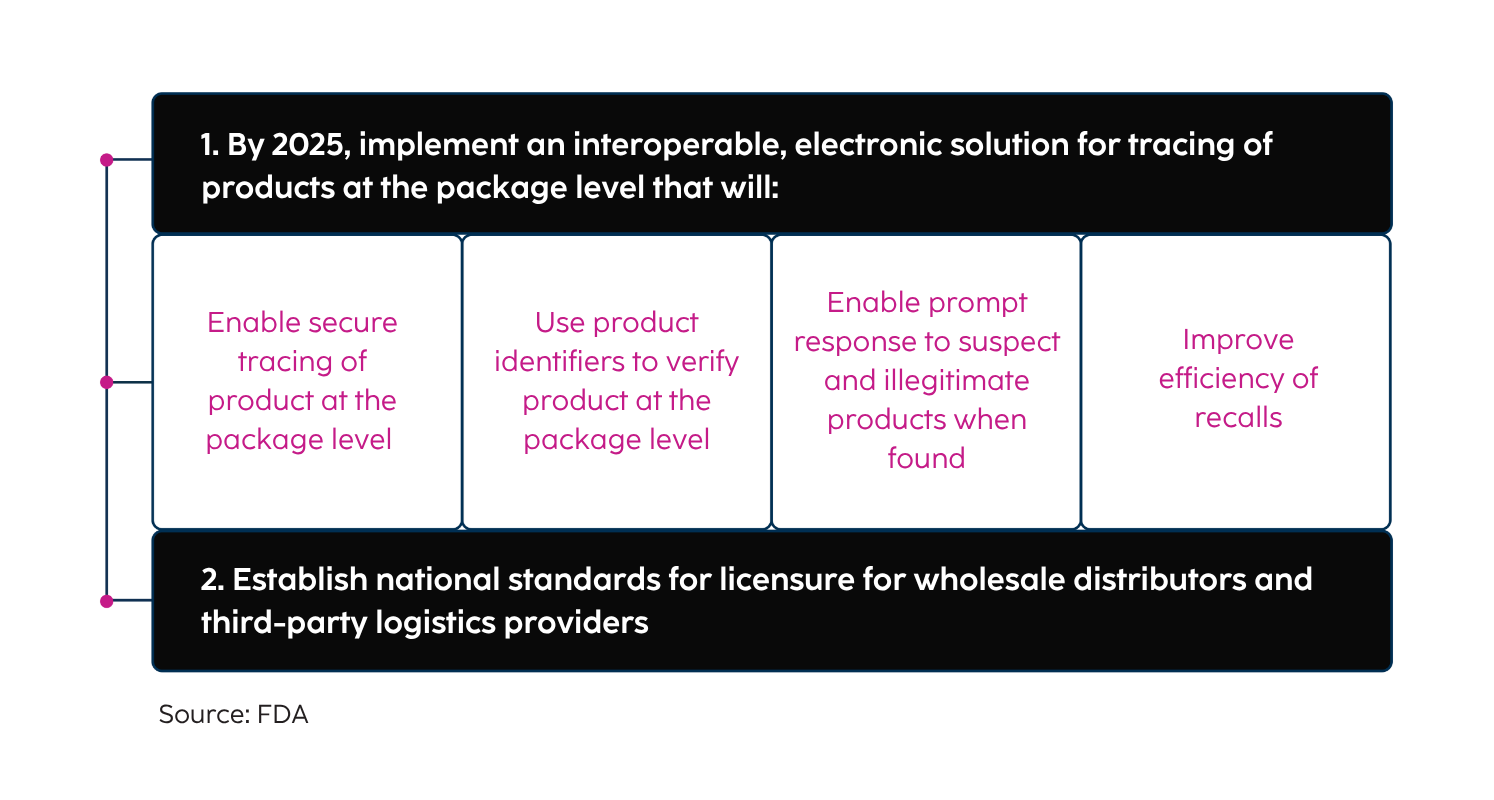
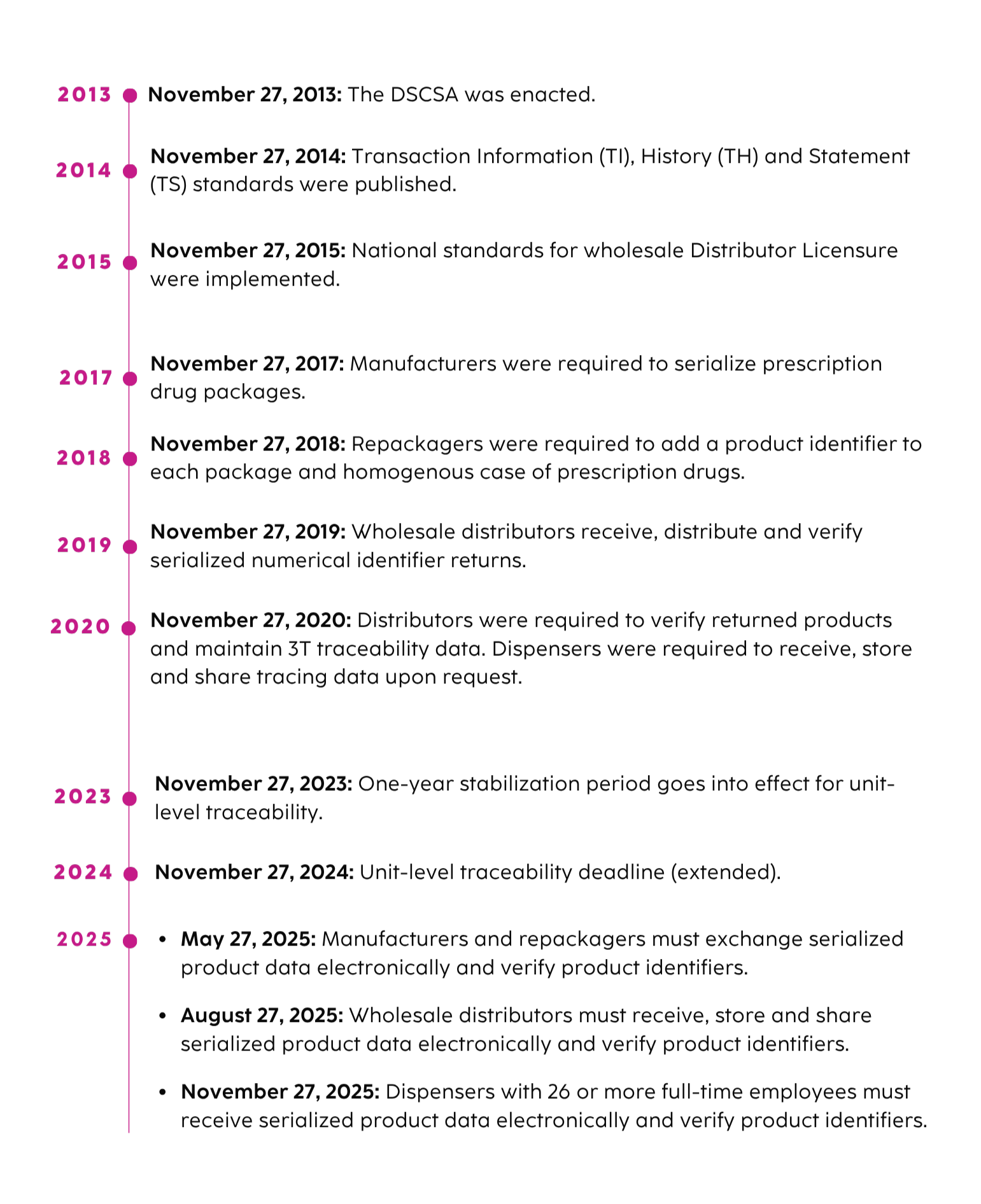
Enacted in 2013, the DSCSA is a U.S. federal law designed to secure the life sciences value chain and protect patient safety by providing a single standard for tracking and tracing certain prescription drugs throughout the supply chain. The law replaces an assortment of state requirements. In response to the growing number of substandard or counterfeit pharmaceutical products, the DSCSA system verifies pharmaceuticals at all stages from the manufacturer to the pharmacist to the consumer. As the phased implementation completed in 2023, full unit-level traceability, including aggregation, is now required.
DSCSA Goals
Benefits of the Drug Supply Chain Security Act (DSCSA)
- Improved patient safety as counterfeit drugs are prevented from entering the supply chain
- New and faster ways to identify illegitimate products in the supply chain
- Safer and more efficient supply chains
- Clearer regulations with a single source of truth
- Stronger distributor licensure standards
DSCSA Milestones & Implementation Timeline
DSCSA Requirements
The DSCSA requires trading partners in the pharmaceutical supply chain – manufacturers, wholesale distributors, repackagers, third-party logistics firms and dispensers (mainly pharmacies) – to track, trace and verify prescription drugs, ensuring that only proper, safe medicines reach patients.
1. Product Identification
Manufacturers and repackagers are required to serialize products by attaching a standardized product identifier that includes a standardized numerical identifier (SNI) unique to each package or case, along with the lot number and expiration date, all presented in both human- and machine-readable formats. This data is embedded in a 2-dimensional data matrix code on each package, and either a linear or 2D matrix barcode on each homogenous case. Additionally, manufacturers and repackagers must maintain records of these product identifiers for a minimum of six years.
2. Product Verification
Manufacturers, wholesale drug distributors, repackagers and some dispensers must provide information that verifies a product is legitimate and must quarantine and investigate a product that might be illegitimate – in other words, a product that is suspected of being counterfeit, diverted or stolen. There must be a process to notify trading partners and the FDA of suspected illegitimate products in a timely manner, and a process for responding to notifications of illegitimate products.
3. Product Serialization
Manufacturers must serialize prescription drugs, including a serial number, lot number, expiration date and product identifier (GTIN® or NDC). Manufacturers must verify products at the package level and must provide 3T documents electronically. Repackagers must ensure serialization for repackaged medicines and must be able to generate their own serial numbers. Repackagers are required to maintain records linking original products to their repackaged versions. They may only handle certain prescription drugs if the products are properly serialized and must perform verification at the package level.
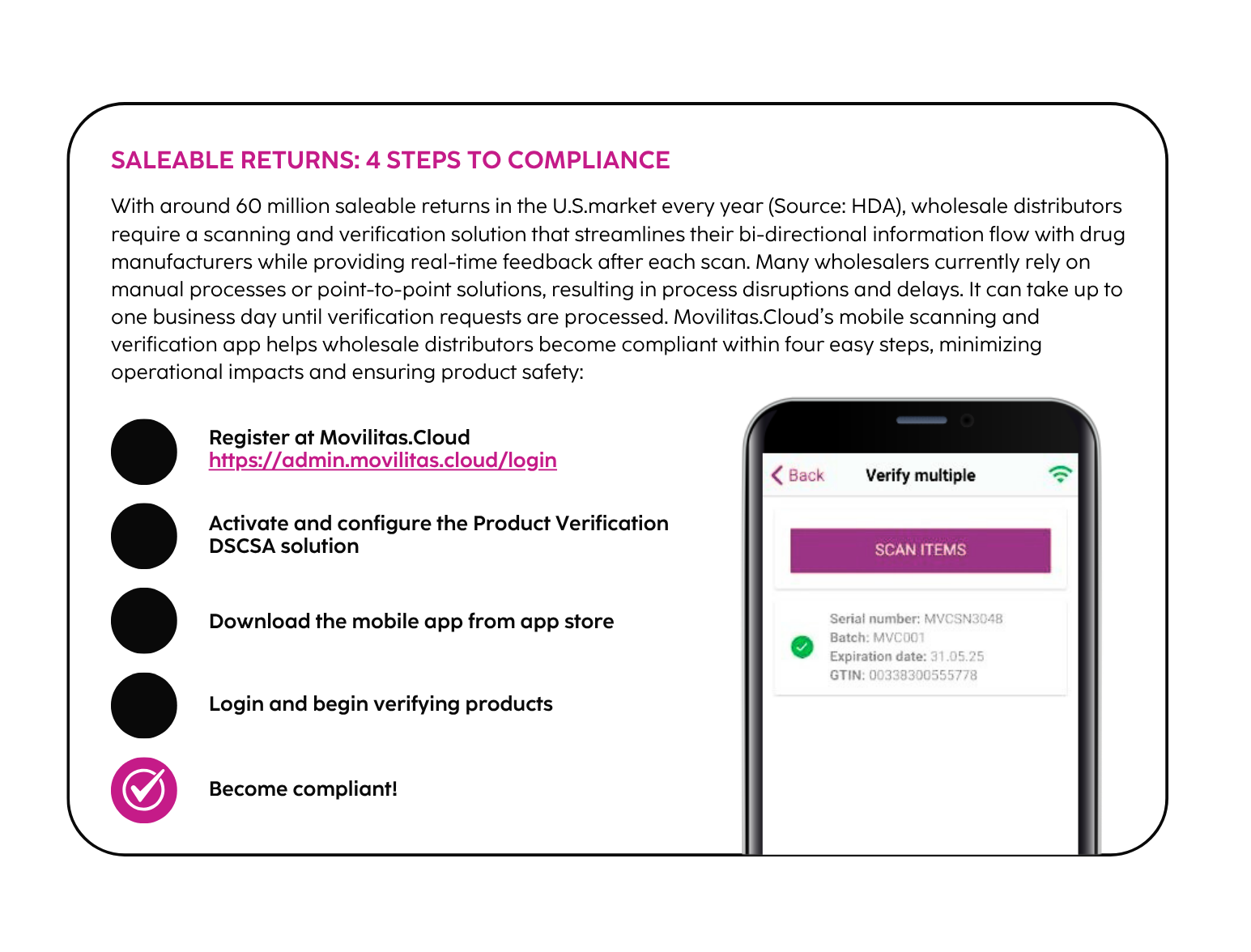
4. Saleable Returns
Wholesale distributors must verify the serialized product identifiers of any saleable returned pharmaceuticals before they can be resold. The wholesaler initiates a verification request to the manufacturer, who must respond within 24 hours with a verification response. Verification includes ensuring the product identifier is correct and true, including the serial number, lot number, expiration date and Global Trade Item Number (GTIN).
5. Authorized Trading Partner (ATP)
As a key part of DSCSA requirements, pharmaceutical dispensers, distributors and manufacturers must only engage with other authorized trading partners. Movilitas.Cloud became an early adopter of this initiative, proving to be a trusted solution partner for fast implementation and credentialing.
6. Product Tracing
Trading partners must provide information about a drug and who handled it for every transaction in the U.S. pharmaceutical market. Manufacturers, wholesale distributors and repackagers, prior to or at the time of the transaction, must provide trading partners with transaction information, transaction history and a transaction statement. Product tracing standards currently require traceability at the lot level, but additional changes took effect when the DSCSA entered the Enhanced Drug Distribution Security (EDDS) phase in 2024, including the requirement to trace products electronically down to the package level.
Vision for the Enhanced Drug Distribution System

What You Need to Do to Comply with the DSCSA
As a manufacturer, wholesale distributor, repackager, third-party logistics firm or dispenser, you have strict responsibilities to your customers and other trading partners, as well as licensing and recordkeeping requirements and a duty to report to the FDA.
1. Trade with Properly Licensed Partners
- Manufacturers and repackagers must have valid FDA registration.
- Distributors and 3PLs must have valid state or federal licenses and must comply with reporting requirements.
- Check the FDA’s Drug Establishment Current Registration Site (DECRS) to verify the licensure of your trading partners.
2. Standardize Operating Procedures to Identify Products that Might be Falsified
- Use form FDA-3911 to notify the FDA within 24 hours if you believe you possess an illegitimate product.
- Notify trading partners within 24 hours.
- Quarantine and investigate products you believe might be illegitimate.
- Take steps to ensure patients do not receive illegitimate product.
3. Share the 3Ts During All Transactions
- Transaction Information (TI)
- Transaction History (TH)
- Transaction Statements (TS)
4. Track and Trace Pharmaceuticals
- Only accept products with complete tracking information, including the 3Ts.
- Generate and provide complete product tracing information when dealing with trading partners.
5. Prepare to Respond to Information Requests
- Respond to requests by trading partners within two business days in the case of a recall or other investigation.
- Store tracing information for at least six years.
6. Check for Proper Product Identifiers
- A product identifier that contains an NDC, serial number, lot number and expiration date in human- and machine-readable format.
- Manufacturers and repackagers must place a product identifier on prescription pharmaceutical packages.
- Dispensers can only buy or sell properly identified and serialized pharmaceuticals.
How We Can Help with Movilitas.Cloud
To comply with the DSCSA and other pharmaceutical regulations, companies must implement robust serialization and reporting processes that connect with both government systems and supply chain partners. Movilitas.Cloud, an intuitive, scalable Software-as-a-Service (SaaS) solution from Engineering Industries eXcellence, helps meet these requirements. Its Serialized Manufacturing and Serialized Logistics applications streamline high-volume serialization, ensure data integration and support end-to-end compliance across the supply chain.