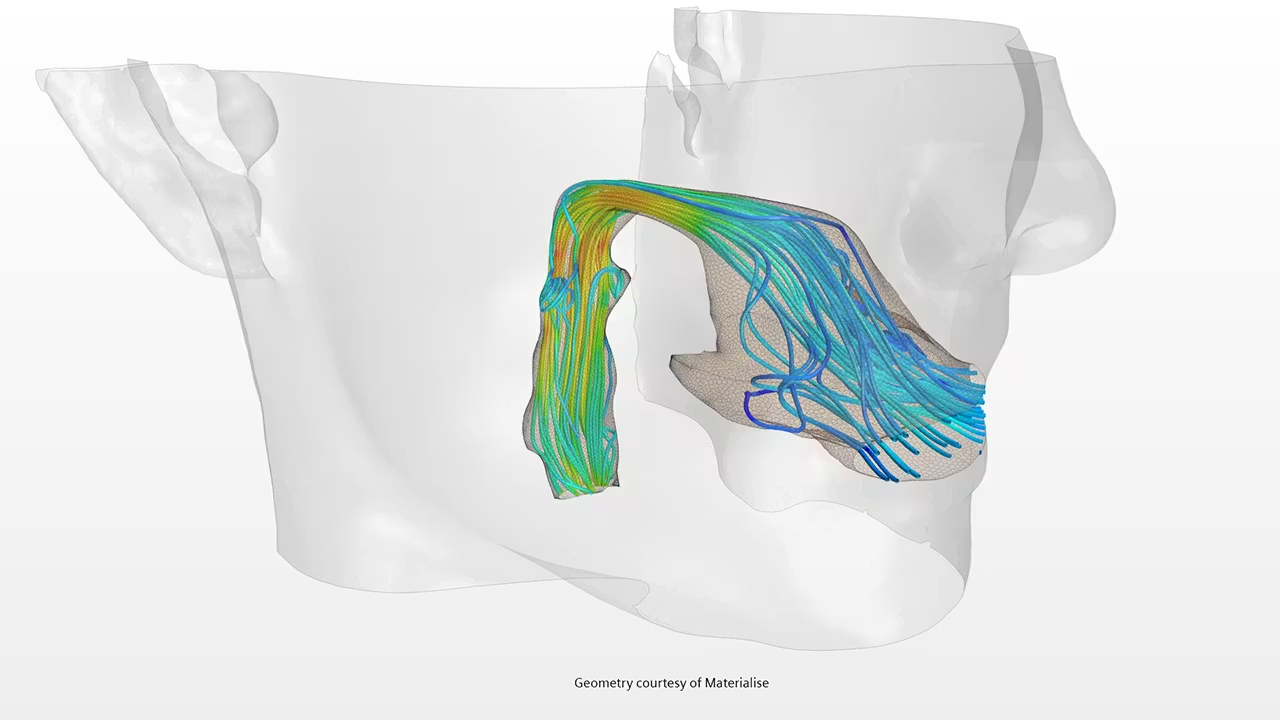
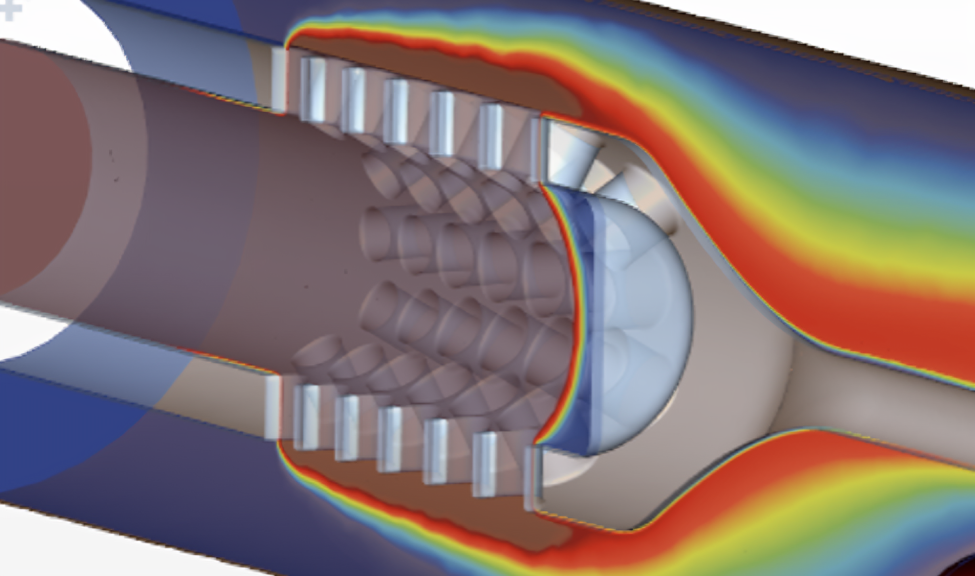
Simcenter STAR-CCM+ for Medical Devices
Your prototype shouldn’t be the first time your device fails
Today, an average of 1.5 medical devices are recalled every week. Every late-stage device failure isn’t just an engineering issue - it’s a financial and regulatory crisis. Clinical trials are more expensive than ever. FDA recalls are climbing across every device category, with design failure as the leading cause. And regulators now expect digital evidence, not guesswork.
Yet teams still discover critical issues only after burning millions in prototyping and trial prep.
A stent collapses under unexpected hemodynamics. An inhaler misdelivers because turbulence wasn’t modeled. An implant overheats only during the third bench test after a year of funding is already gone.
These aren’t surprises - they’re the cost of not using multi-physics simulation early enough or deeply enough.
This is where IndX changes the economics of medtech development.
We turn Simcenter STAR-CCM+ into your digital validation engine, revealing the physics that cause recalls, derail trials, blow budgets, and sink timelines. One environment. 1000s of designs analyzed.
With IndX and Simcenter STAR-CCM+, your funding lasts longer, your designs get safer, and your time-to-market gets shorter.

Why IndX & Simcenter STAR-CCM+ for Medical Devices
At IndX, our experts bring years of CAE simulation, design optimization and medical devices expertise to solve your biggest challenges with medical device development.
Problem: Slow, expensive prototype cycles
Solution: Virtual multi-physics studies that cut iterations 35-50%.
Problem: Hidden failure modes engineers can’t see early
Solution: CFD + FEA workflows capturing real blood, air, heat, stress.
Problem: Fragmented R&D slowing FDA/MDR evidence creation
Solution: Integrated design-to-simulation loop with traceable validation outputs.
Problem: Limited in-house simulation resources
Solution: IndX experts + device templates accelerate execution immediately.
Problem: Burning cash and runway on preventable redesigns
Solution: Early failure discovery that protects budget, timeline, and investor confidence.
Build Better Medical Devices, Faster
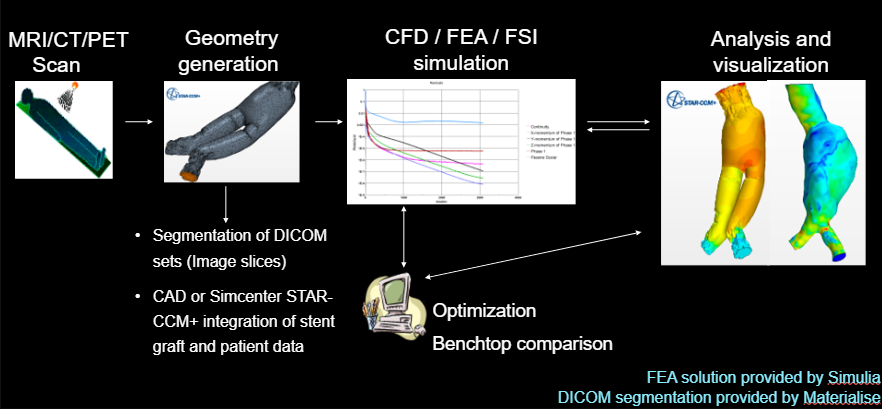
We’ve helped 19 out of top 20 leading pharma companies, surgical device OEMs, medical equipment manufacturers, wearable device companies and others get to market faster and safer.
How IndX works with Medical Device companies
We offer a no-cost, expert-led assessment covering:
- Review of your current CFD/FEA workflow
- Identification of bottlenecks and accuracy issues
- Recommendations to accelerate verification cycles
- STAR-CCM+ multiphysics opportunities
- Automation & integration roadmap
- Regulatory traceability improvements
Our free assessment is perfect for R&D, CFD/FEA, V&V, and Simulation teams. Whether you are just starting your simulation journey or have an established simulation practice, we can help with expert medical device and simulation consultants.
Terumo Corporation - Designing efficient blood pumps with Simcenter STAR-CCM+
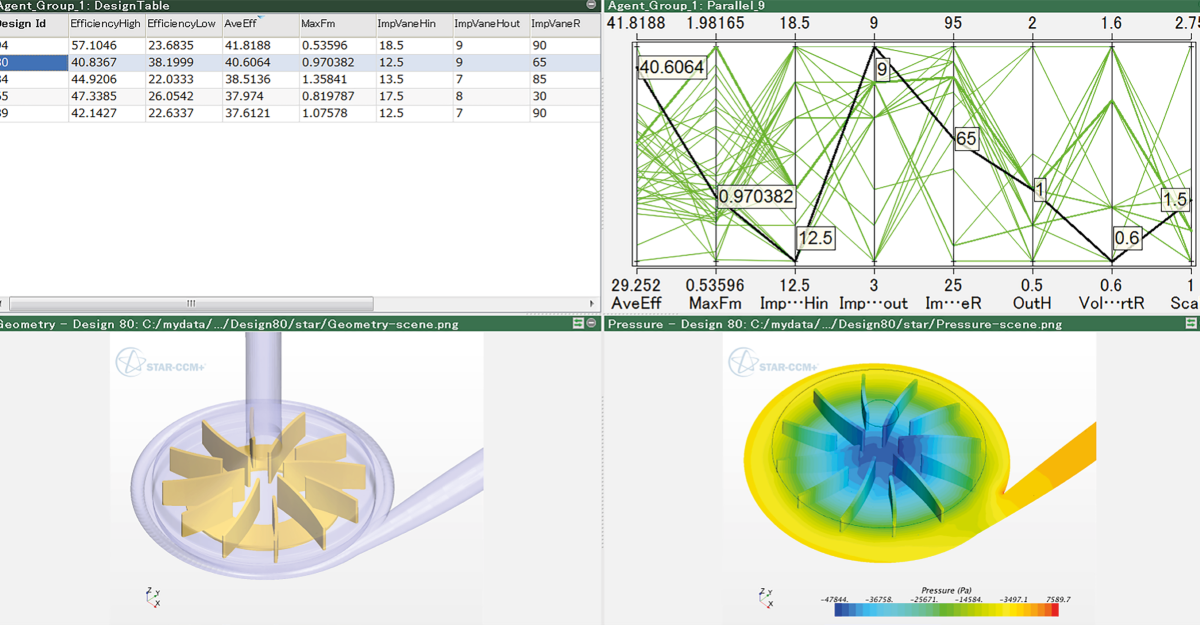

B&W Engineering – Improving peristaltic pump efficiency to reduce prototype cycles
- Challenge: Designing a life-sustaining peristaltic pump demanded precise, constant flow and strict safety margins under complex conditions. But traditional prototype-first development was slow, risky, and costly (each prototype could cost ~€80,000).
- Solution: Using Simcenter STAR-CCM+, B&W Engineering simulated fluid, structural, and thermal behavior including fluid-structure interactions and hyper-elastic tubing deformation. This refined the pump geometry virtually, cut prototype cycles, and deliver a design with 3% improved drug-delivery accuracy before building a single physical unit.
Why Medical Device companies choose Simcenter STAR-CCM+
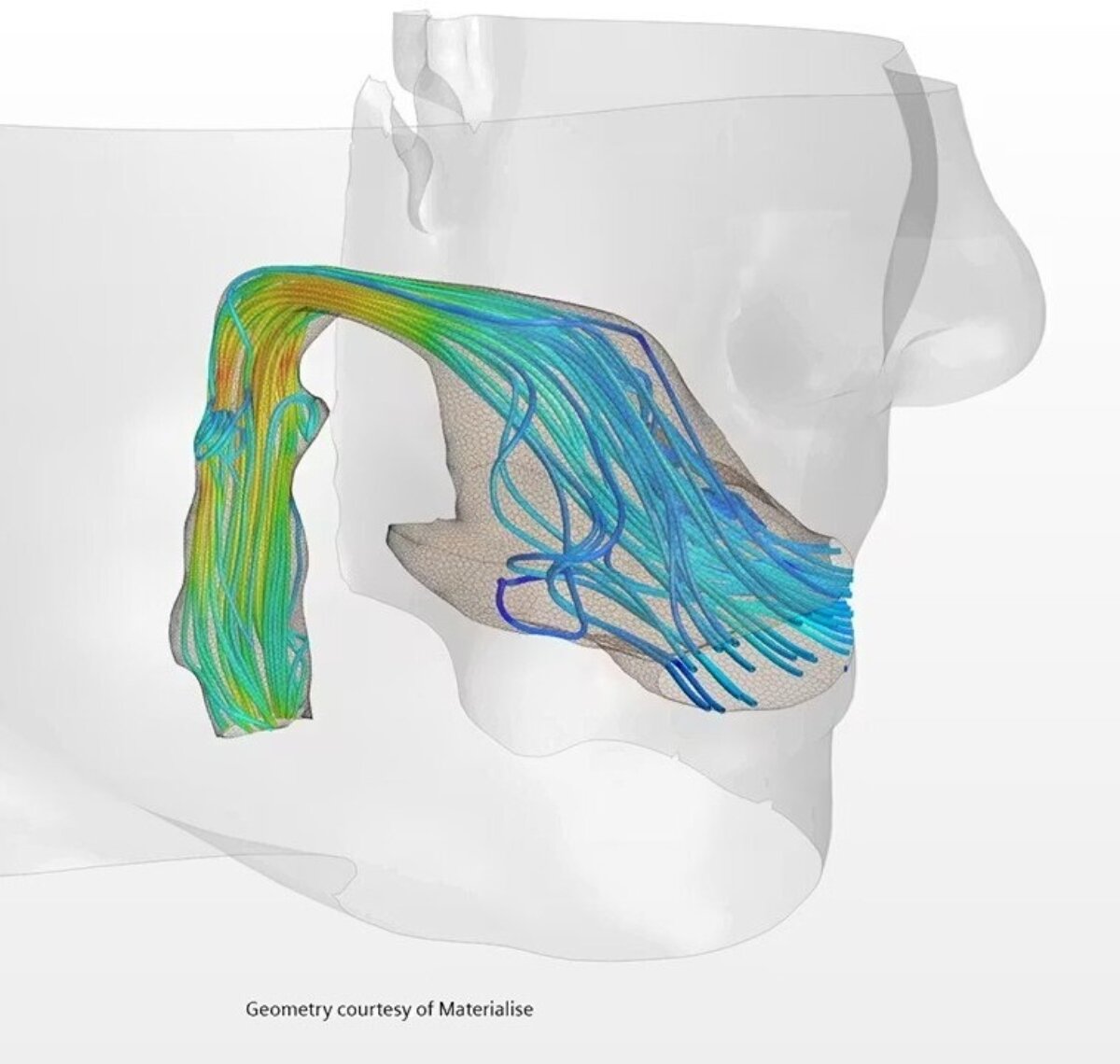
Simcenter STAR-CCM+ captures the full spectrum of behaviors that determine device safety, reliability, and durability:
-
Hemodynamics: stents, valves, catheters
-
Aerosol & airflow: inhalers, ventilators, airway devices
-
Thermal behavior: implants, wearables, surgical tools
-
Structural loads & fatigue: durability and lifetime prediction
-
Particle deposition: drug delivery pathways, contamination models
-
Scale-up challenges: pumps, mixers, reactors, channels
You discover problems when they cost the least - not during trials when the stakes are highest.
IndX helps you overcome medical device design challenges with our team of world-class consultants and experts.
Outcomes we deliver
- 35-50% reduction in prototype cycles
- 2-4x faster design iterations
- Significant reduction in design-failure risk
- Regulator-ready simulation evidence
- Earlier discovery of failure modes that drive recalls
- Improved durability, safety, and reliability
- Reduced clinical trial cost and complexity
Ready to eliminate device failure before it's expensive?
We help you design safer, faster, more resilient medical devices together backed by real physics, real evidence, and real execution.